04-Crystallography
Summary
In solid form, many molecules line up in regular arrays showing long range order. These are crystals. Classically these are distinguished by flat faces and sharp edges, but in reality most solids are crystalline, just that the individual crystals are so small we don’t recognise this.
The experimental study of crystals is a powerful way to unpick the structure of the molecules from which they are composed.
Contents
- What are crystals?
- Why are we interested?
- Where do we get information?
- Unit Cells
- Bravais Lattices
- BCC / FCC / HCP Crystals
- Layers & Stacking
- Positions and Planes in Crystals
- Examples of Different Crystal Types
What is a Crystal?
- Arrangement of atoms with repeated pattern in three dimensions.
- Array of points in the crystal look identical (same surrounding atoms, same orientation)
- Examples:
- gems (diamond, ruby)
- ceramics (silicates, alumina)
- metals (iron, coppe)
- even polymers.
- The opposite of a crystal is an amorphous or glassy solid. On an atomic scale these looks like frozen liquids.
- Can be tough to spot from external appearance
- e.g. a smooth shaped piece of glass is NOT a crystal;
- e.g. A rough irregularly shaped particle may consist of crystalline material.
Metals have Crystals
- Image below shows crystal grains in copper
- Polish and etch to reveal grain boundaries

Why do we care?
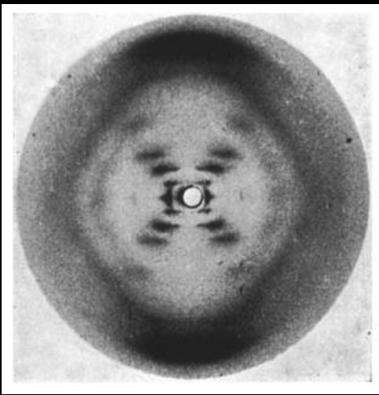
Information on Crystals
- Cambridge Structural Database (CSD):
- Information on >1 million crystals (1046700, March 2020)
- Nomenclature (how to name them)
- Freely available
- http://www.ccdc.cam.ac.uk/
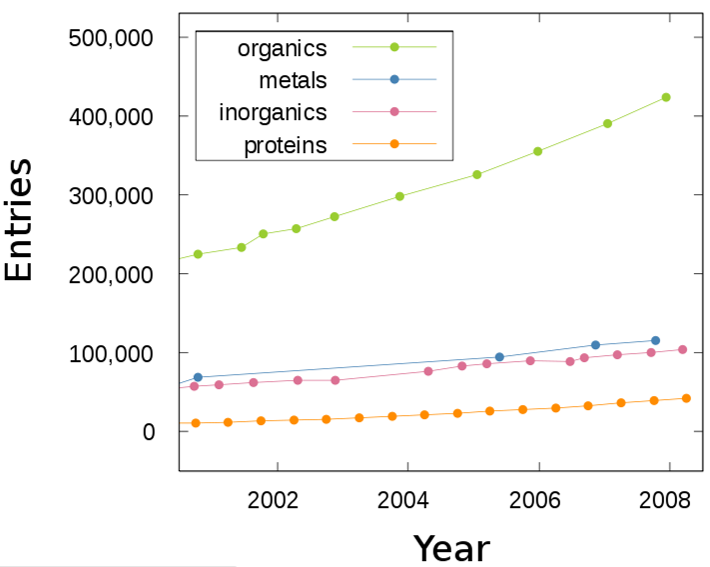
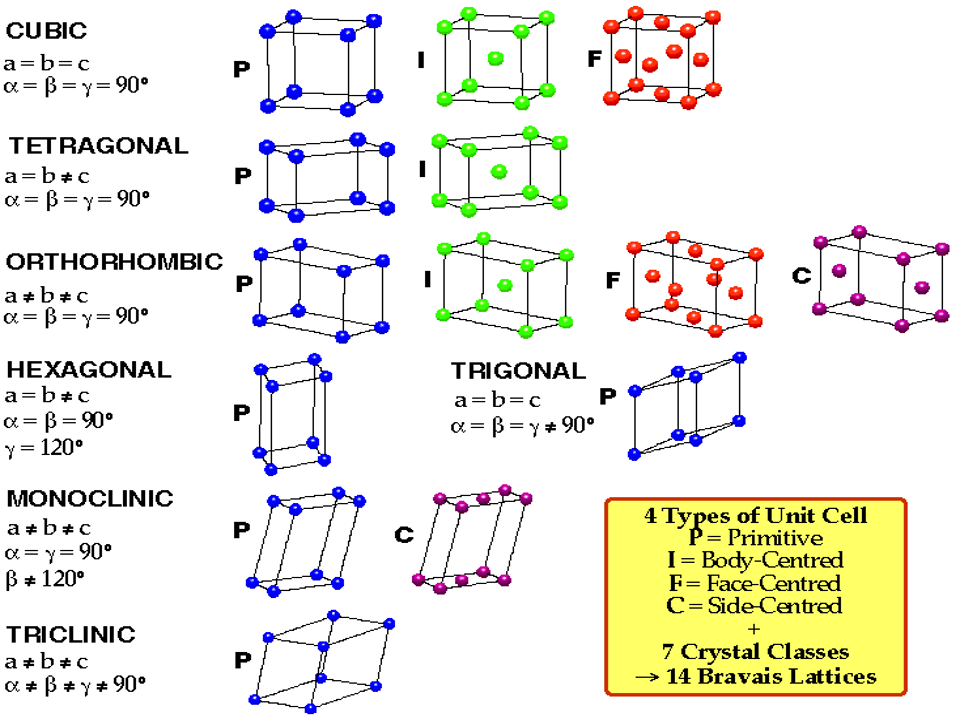
Unit Cells
- Fundamental building blocks of crystals
- Arrangement of blocks must fill all space
- Seven different shapes of unit cell allowed namely;
- cubic
- tetragonal
- rhombohedral
- hexagonal
- orthorhombic
- monoclinic
- triclinic
Parameters for Unit Cells
- cubic: \(a\;=\;b\;=\;c\;\) and \(\;\alpha\;=\;\beta\;=\;\gamma\;=\;90^{\circ}\)
- tetragonal: \(a\;=\;b\;\neq\;c\;\)and \(\;\alpha\;=\;\beta\;=\;\gamma\;=\;90^{\circ}\)
- orthorhombic: \(a\;\neq\;b\;\neq\;c\;\) and \(\;\alpha\;=\;\beta\;=\;\gamma\;=\;90^{\circ}\)
- rhombohedral: \(a\;=\;b\;=\;c\;\) and \(\;\alpha\;=\;\beta\;=\;\gamma\;\neq\;90^{\circ}\)
- hexagonal: \(a\;=\;b\;=\;c\) and \(\alpha\;=\;\beta\;=\;90^{\circ}, \gamma\;=\;120^{\circ}\)
- monoclinic; \(a\;\neq\;b\;\neq\;c\;\) and \(\;\alpha\;=\;\beta\;=\;90^{\circ}\;\neq\;\gamma\)
- triclinic: \(a\;\neq\;b\neq\;c\;\) and \(\;\alpha\;\neq\;\beta\;\neq\;\gamma\;\neq\;90^{\circ}\)
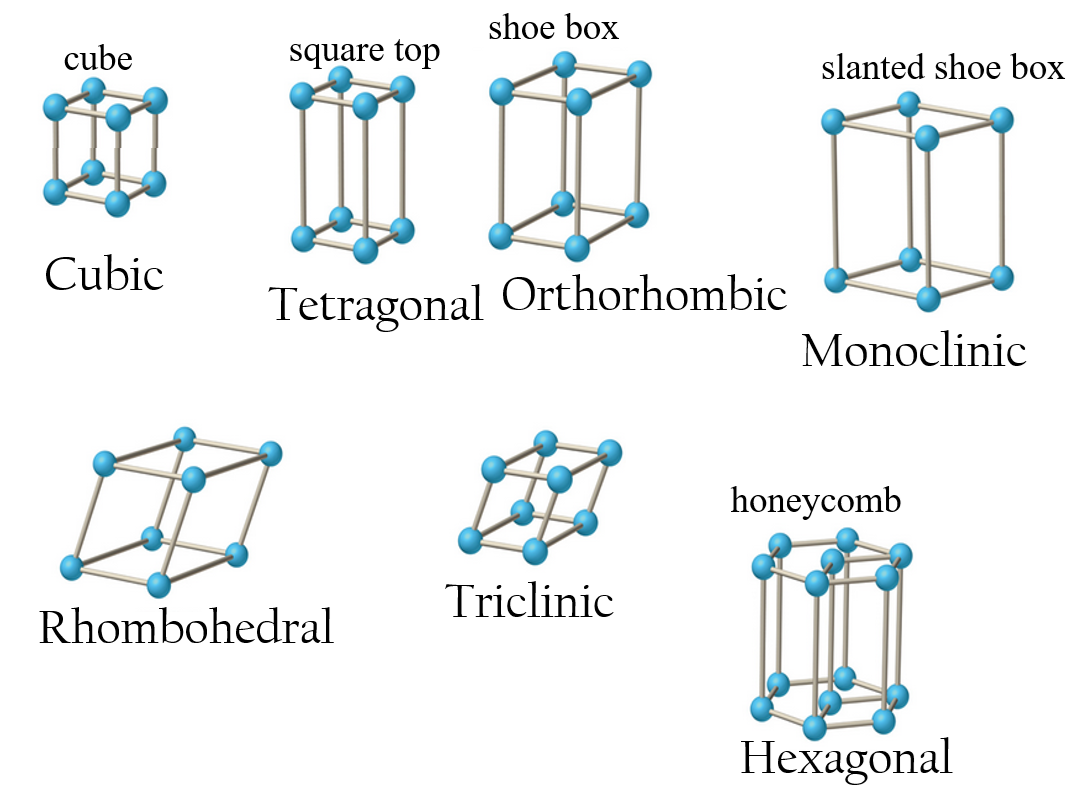
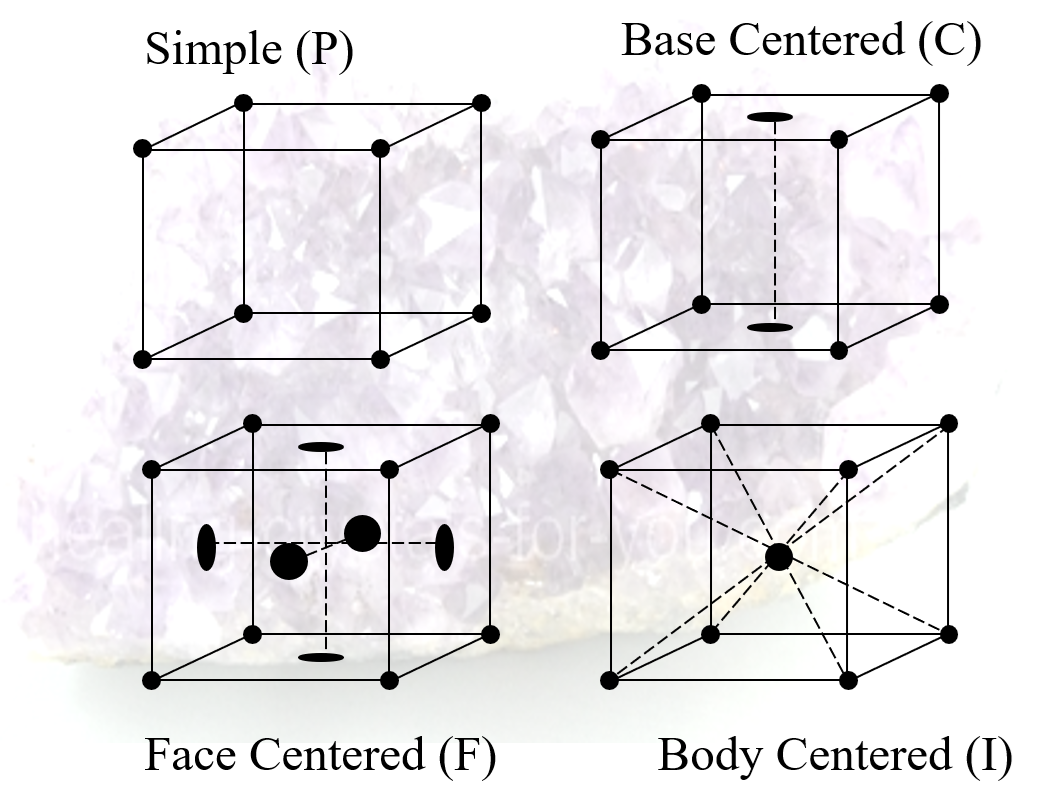
Symmetries within Unit Cells
- As well as the symetries of their shapes, unit cells can have symmetries due to their internal structures
- Gives four types of unit cell
- simple (P)
- face centered (F)
- body centered (I)
- base centered (C)
Bravais Lattices
- Not all of the seven cell shapes can have all four types of internal symmetry
- For instance, the face centered tetragonal structure can be reduced to the body centered tetragonal structure
- There are fourteen distinct possibilities called the Fourteen Bravais lattices
- The fourteen lattices contain all the necessary translational symmetries to describe every possible crystal structure in nature
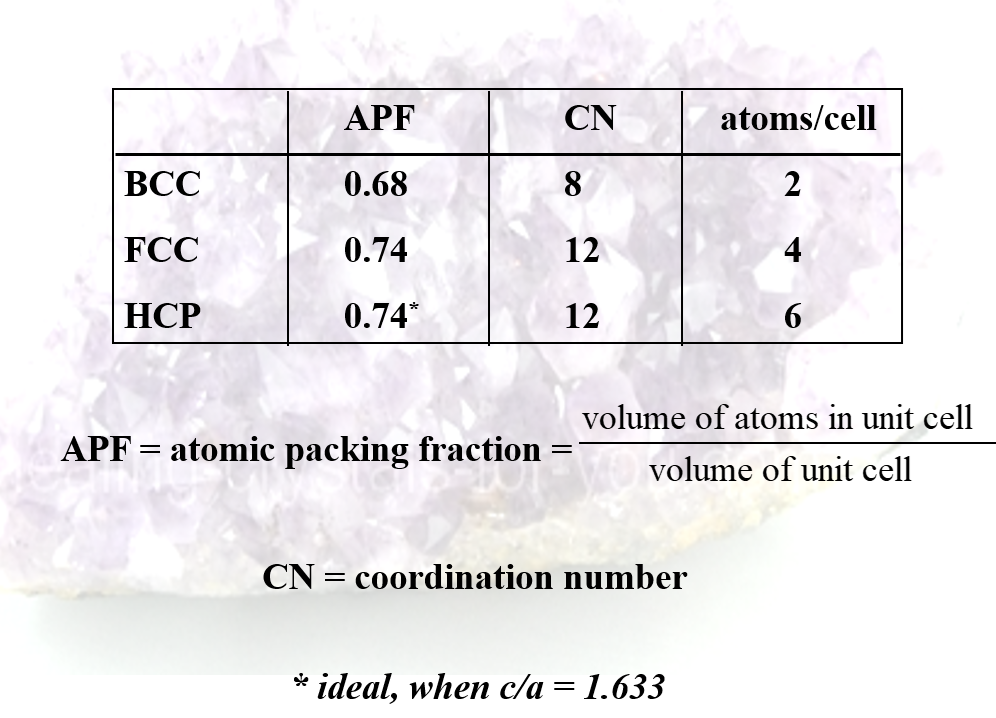
Metallic Crystal Structures
- Because of the nature of the metallic bond the principle requirement is for a high density
- Three arrangements satisfy this criterion
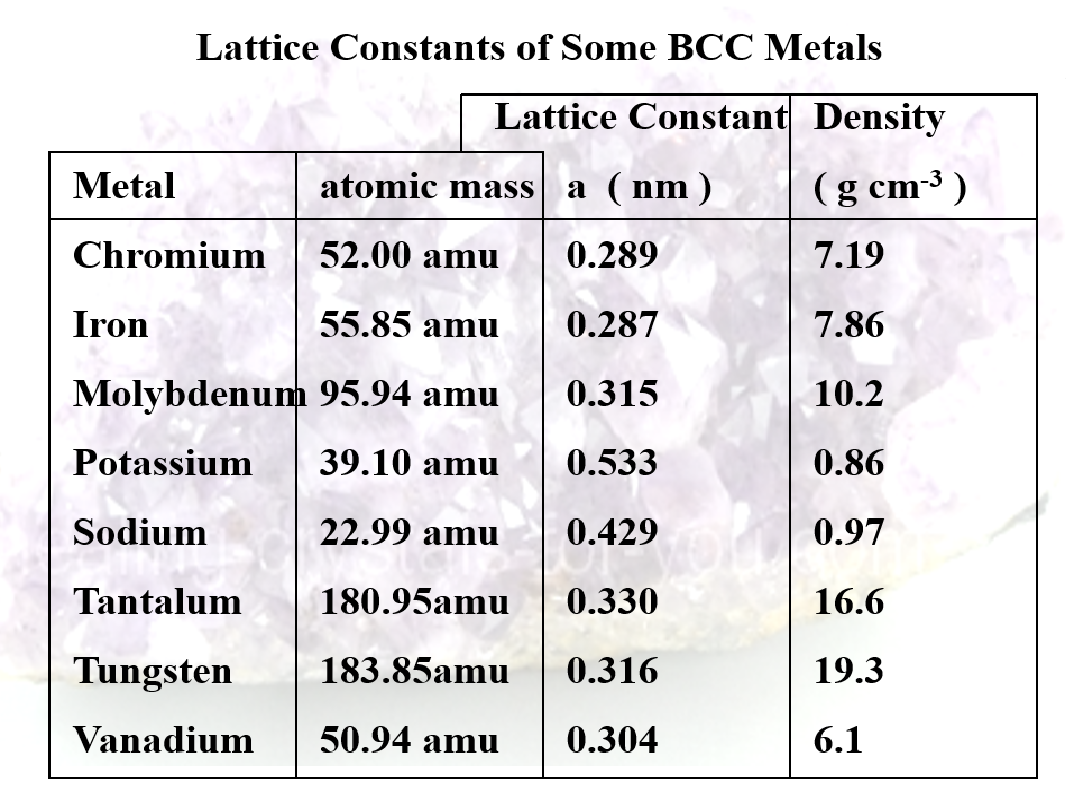
- body centered cubic (BCC)
- face centered cubic (FCC)
- hexagonal close packed (HCP)
Body Centred Cubic, Hexagonal Close Packed, and Face Centred Cubic
Layers and Stacking
- We’ll talk about metals here
- not so fussy about direction of bonds
- behave like hard spheres
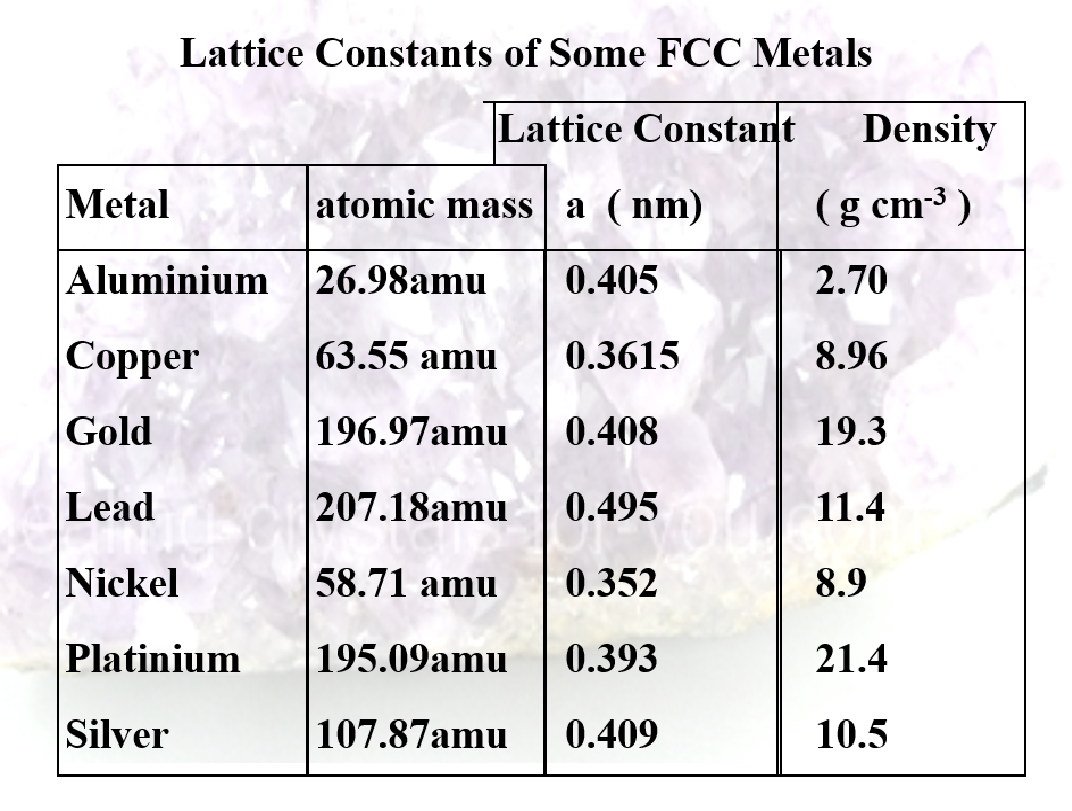
- Lay spheres out on a table so they are packed as close as possible in one layer
- Two possibilities for next layer
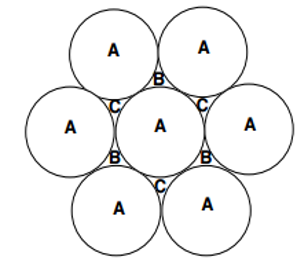
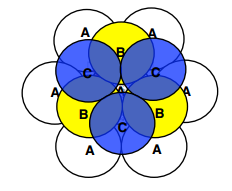
- There are two ways to stack layers
- ABABABABA
- Creates form called Hexagonal Close Packed (HCP)
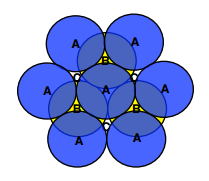
- ABCABCABC
- Creates form called Face Centred Cubic(FCC)
Positions, Directions, and Planes
- Position
- specifies a point within a unit cell
- expressed as fractions of lattice vectors.
- notation is ( 1/4, 0, -21/2 )
- Direction
- specifies the direction of a line
- expressed as a set of integers
- notation is [210] where 1 means -1.
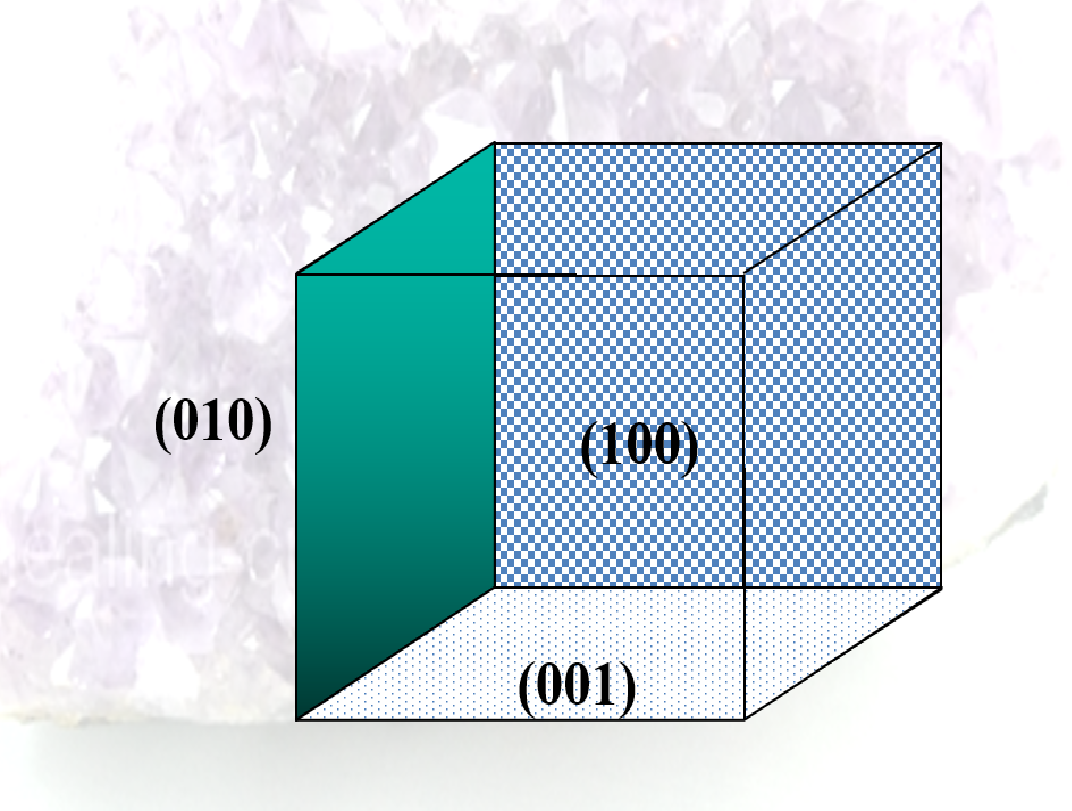
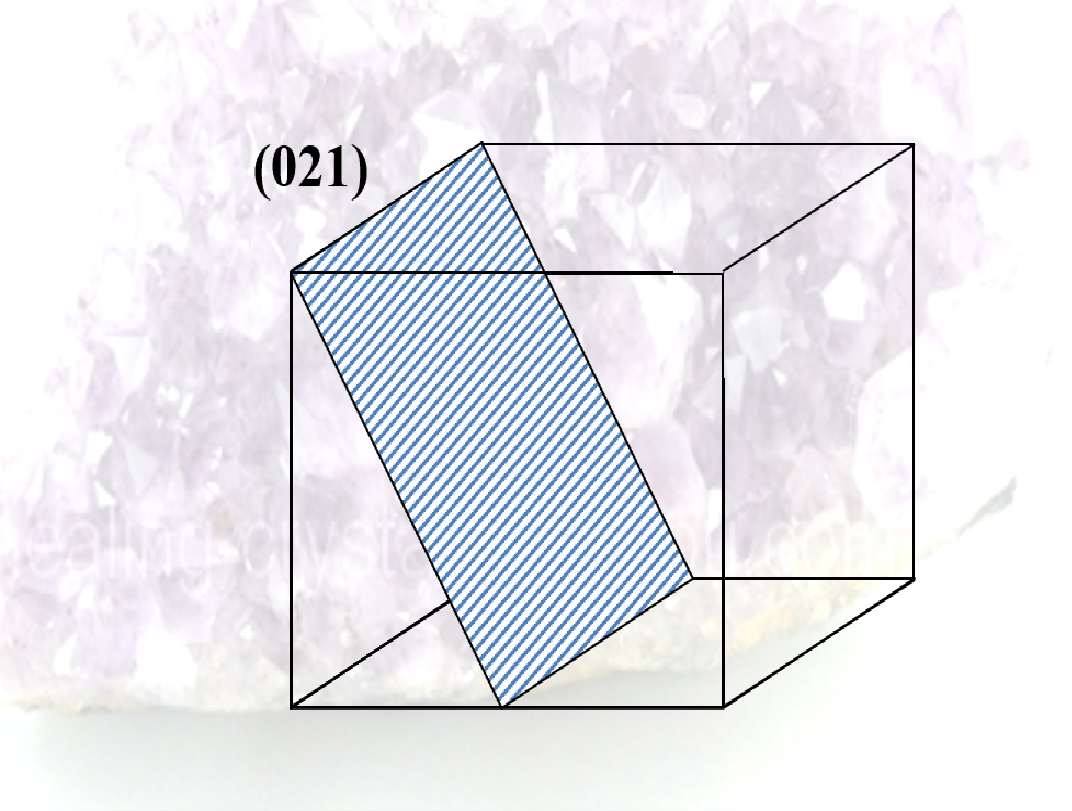
- Planes
- specifies surfaces within the crystal
- expressed as a set of integers known as the Miller indices
- notation is (210).
Hints for Positions
- by convention, take the position (0,0,0) to be in the bottom left hand corner at the back
- remember which axes point in which directions.
- positions can be fractions; 21/2 , -3/4 , 8/3 , etc
- remember the notation; (x,y,z)
- positions with negative indices or indices >1 will be outside the unit cell
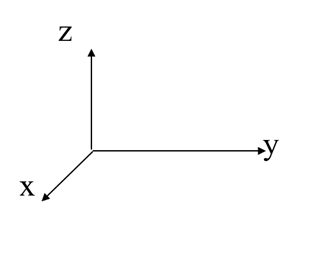
Hints for Planes
- sets of parallel planes all have the same Miller indices
- choose one of the planes that does not pass through the point (0,0,0)
- see where the plane cuts the x, y, and z axes (the intercepts, could be infinity)
- get the reciprocals of these intercepts
- clear fractions
References

Physics - Quantum