01-Radiation
Summary
Radioactivity is the spontaneous emission of high energy particles due to the decay of unstable nuclei
Contents
- types of radiation
- sources of radiation
- applications of radioactivity
- safety aspects of radioactivity
- radioactive decay laws / radioactive half life
- measurement of radiation / units radiation limits
| Summary of Properties of Radiation | |||
|---|---|---|---|
| α | β | γ | |
| nature | 2 protons + 2 neutrons | electron | light ray |
| effects of em fields | weakly afected | strong | no effect |
| penetration | cm's in air, sheet of paper | mm's of aluminium | cm's of lead |
| ionise atoms | strongly | weakly | very weakly |
| speed | 10% speed of light | 50% light | speed of light |
| typical energy | 5MeV | 1MeV | 100keV |
| detectors | photo film | G-M tube | G-M tube |
Notation
Syntax
\(^A_ZElement\;e.g.\;^4_2He\;has\; A=4\; and\; Z=2\; (sometimes\; just\;^4He)\)
radioactivity originates in the nucleus. We need to develop a notation to discribe the processes in the nucleus.
Atomic Number ( Z ) is the number of protons in the nucleus.
Atomic Mass ( A ) is the combined number of protons and neutrons in the nucleus.
Isotopes are nuclei with the same number of protons but different numbers of neutrons
Nucleus emits an \(\alpha\) particle
- loses two protons and two neutrons
- Z decreases by 2 and A decreases by 4
- \(^A_ZParent\;=>\;^{A-4}_{Z-2}Daughter\;+\;\alpha\)
- examples
- \(^{238}_{92}U\;=>\;^{234}_{90}Th\;+\;\alpha\)
- \(^{210}_{84}Po\;=>\;^{206}_{82}Pb\;+\;\alpha\)
Nucleus emits a \(\beta\) particle
- neutron in the nucleus splits into a proton and an electron
- electron emitted as a beta particle
- Z increases by 1 and A remains the same
- \(^A_DP\;=>\;^{A}_{Z+1}D\;+\;\beta \; + \; \nu\)
- examples
- \(^{99}_{42}Mo\;=>\;^{99}_{43}Tc\;+\;\beta \; + \; \nu\)
- \(^{14}_{6}C\;=>\;^{14}_{7}N\;+\;\beta \; + \; \nu\)
Nucleus emits an \(\gamma\) ray
- no change in A, Z, just loss of excess energy from nucleus
Sources of Radiation
\(\alpha\) particles
- almost all alpha particles originate in the radioactive decay of heavy nuclei e.g. \(^{238}U\), \(^{226}Ra\), etc.
- \(^{208}Pb\) is the heaviest stable nucleus known.
- typically \(\alpha\) energy of 4MeV (\(6.4 \times 10^{-13}\) Joules)
- ~highest is \(^{212}_{84}Po\; \rightarrow \; ^{208}_{82}Pb \;+\; \alpha\;+\;8.78MeV\)
\(\beta\) particles
- naturally occuring sources of beta particles are rare.
- They can be prepared by irradiating target nuclei with neutrons ( e.g. \(^{125}I\) for thryoid treatments ).
- The beta emitter \(^{14}C\) is produced by cosmic rays in the upper atmosphere
- bigger range of energies than \(\alpha\) particles
- 3.5MeV for \(^{137}_{54}Xe \; \rightarrow \; ^{137}_{55}Cs \;+ \; \beta \; + \; \nu\)
- 18keV for \(^{3}_{1}T \; \rightarrow \; ^{3}_{2}He \;+ \; \beta \; + \; \nu\)
\(\gamma\) rays
- after \(\alpha\) or \(\beta\) decay, the daughter nucleus contains excess energy.
- This is liberated by the release of a \(\gamma\) ray
- about 100keV typical energy
Applications of Radiation
Medicine;
- diagnostic imaging: radioactive tracers (\(^{99}Tc\)) latch onto a particular chemical in the body. The distribution of this chemical can be followed by radiation detectors
- PET scans use \(^{18}F\) tracers to detect cancers.
- radiotherapy: focus radiation in the diseased part of the body, healthy cells recover, diseased cells die. \(^{131}I\) in treatment of thyroid disorders.
Appliances:
- devices such as smoke detectors use radiation (\(\alpha\) particles from \(^{241}Am\))
Archeology:
- radioactive dating; in dead material the ratio of \(^{14}C\) to \(^{12}C\) determines age.
- \(^{14}C\) has a useful half-life of 5730 years
- Works for aging rocks as well; ratio of Uranium to Lead.
Engineering;
- checking welds
- thickness control
- testing wear and tear of a machine
- leak detection
- sterilising food
Radiation Safety
- radiation damages tissue
- produces reactive chemicals (radicals) in the body
- \(\alpha\) particles are the most dangerous, considered 20 times more dangerous than \(\beta\)’s
- because they all their energy in a smaller volume
- \(\gamma\) rays are more difficult to control because of their greater penetration
Radon
- large dose of radiation due to the build up of radon in houses
- radon decays to polonium, bismuth, etc. These latch onto dust particles and can be absorbed by the lungs. They emit a cascade of alpha particles
- 20,000 deaths (lung cancer) per year in the USA due to radon
- radon dose determined by:
- amount of uranium in underlying rocks
- permeability of topsoil
- cracks and fissures in floor
- ventilation in house
- buildings from 1980’s on
- fine, contain impermeable plastic layer in foundations
- buildings from before 1950’s
- fine, because so draughty
Radioactive Decay Laws
- decay of individual nucleus is a totally random process, occurs with certain probability in a certain time period
- for large numbers of nuclei can use statistics
- rate of disintegrations from a nuclear source is proportional to the number of radioactive nuclei present
Mathematics of Radioactive exponential decay
- \(-\frac{dN}{dt}\;\alpha\;N\)
- \(-\frac{dN}{dt}\;=\;\lambda\;N\)
- \(N\;=\;N_0e^{-\lambda t}\)
- where N is the number of radioactive nuclei present at a particular time
- \(-\frac{dN}{dt}\) is the rate of decrease in N.
- \(N_o\) is the original number of nuclei present.
- \(\lambda\) is called the radioactive decay constant and depends on the nature of the nucleus
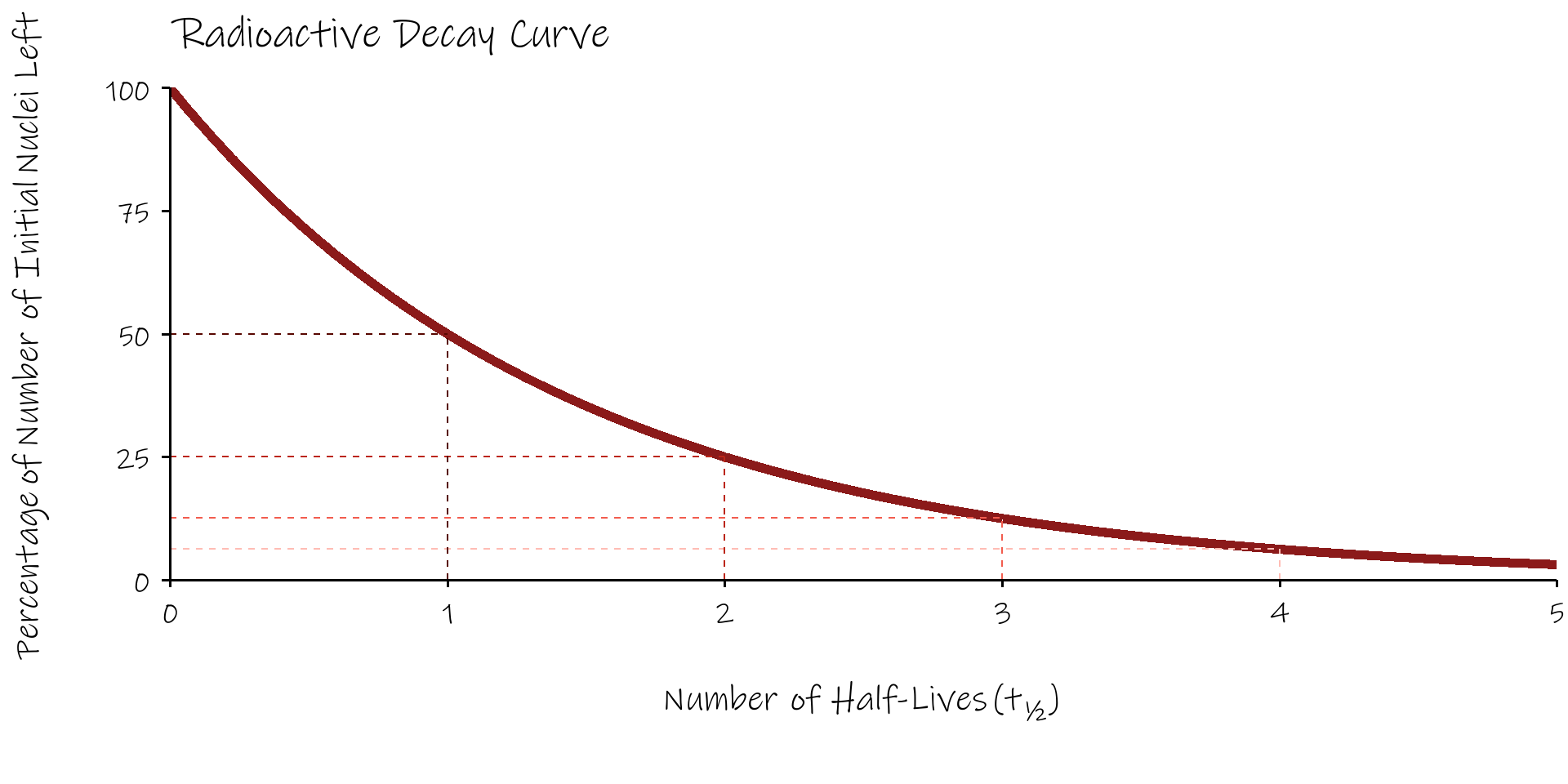
Radioactive Half Life, \(t_{1/2}\)
- a convenient way of discribing radioactive decay is in terms of the radioactive half life, \(t_{1/2}\) .
- the half life is defined as the time taken for the number of radioactive nuclei present in a source to fall to half it’s original value.
- half lives of naturally occurring radioisotopes vary over a wide range
- \(3 \times 10^{-7}\)s (\(^{212}Po\)) to \(1.4 \times 10^{10}\)years (\(^{232}Th\))
the half life is related to the decay constant by:
- \(t_{1/2}\;=\;\frac{log_e2}{\lambda}\;=\;\frac{0.693}{\lambda}\)
Radioactive Decay Curve
Measurement of Radiation
Activity
- This is the number of disintegrations occurring per second.
- It is easily measured with a Geiger counter.
- This activity is proportional to the number of radioactive atoms in the sample.
- \(Activity\;=\; -\frac{dN}{dt}\; =\;\lambda N\)
The units of activity are:
- Curie (Ci)
- \(1\;Ci\; =\; 3.7 \times10^{10}\; disintegrations/sec\)
- A clinical source of \(^{60}Co\) has an activity of several Ci
- An internally administered dose for cancer treatment would have an activity of \(10^{-3}\) Ci
- Becquerel (Bq)
- 1 Bq = 1.0 disintegrations /sec
- This is the SI. unit (1 Ci = \(3.7 \times 10^{10}\) Bq)
Absorbed Dose
- This is the effect of the radiation on the absorbing material
- The SI unit for absorbed dose is the gray (Gy) which is the amount of radiation which deposits energy at a rate of 1 J / kg in any absorbing material.
- (The old unit was the rad … 1 Gy = 100 rad)
- The absorbed dose depends on
- the strength of a given radiation beam (number of particles per second)
- the energy per particle but also on the type of material absorbing the radiation
- This is not the most meaningful units for measuring the biological damage produced by radiation.
- This is because equal doses of different types of radiation cause differing amounts of damage (1 gray of \(\alpha\) particles produces 10 to 20 times the damage of 1 gray of \(\beta\) particles or \(\gamma\) rays)
- This is because the \(\alpha\) particles move more slowly than \(\beta\) and \(\gamma\) rays of equal energy due to their greater mass and ionising collisions occur closer together resulting in more irreparable damage
- The relative biological effectiveness (RBE) or quality factor (QF) of a given type of radiation is defined as the number of grays of X-ray or \(\gamma\) radiation that produces the same biological damage as 1 gray of the given radiation.
- The product of the dose in grays and the QF gives a unit of sieverts (previously rem)
- 1 sievert of any type of radiation does the same approximate biological damage
- Radioactive background 1.3 mSv /year
- X-rays 0.7 mSv / year
Exposure Limits
- 5 mSv per year exposure to the general population
- 50 mSv per year to nuclear workers
- A 4 Sv dose in a short interval of time is fatal with 50% of cases but when spread over a few weeks it is not usually fatal.
- A 10 Sv dose is nearly always fatal
Principle sources of radiation
- Radon - 1mSv per year
- CT scans - 0.8mSv per year
- Nuclear Medcine - 0.4mSv per year
- Cosmic Rays - 0.15mSv per year
- internal radiation (mostly potassium, \(^{40}K\)) 0.03mSv per year
- get total of about \(10\mu Sv\) per day. Below legal limit.
Equations
\(N\;=\;N_0e^{-\lambda t}\)
\(t_{1/2}\;=\;\frac{log_e2}{\lambda}\;=\;\frac{0.693}{\lambda}\)
\(Bq = Ci \times 3.7\times10^{10}\)
\((energy\;in\;joules) = MeV \times 1.6\times 10^{-13}\)
\(Gray = \frac{Bq \times (energy\;in\;joules)}{(person\;mass\; in \;kg)}\)
\(Sv = \frac{Bq \times (energy\;in\;joules) \times RBE }{(person\;mass\; in \;kg)}\)
\((expected\;number\; of\; deaths) = \frac{(dose\; in\; Sievert) \times (population)}{50}\)
typical daily dose = 10\(\mu\)Sv.
\(LD_{50 / 30}\) = 4Sv
References

Physics - Quantum